轻蜂加速器
匿名浏览,不显示本地IP

无论您身在何处,都可像在家上网

全球62个国家中的数百台高速服务器
轻蜂加速app下载受到世界各国地区朋友的的信赖

为什么要选择轻蜂加速器

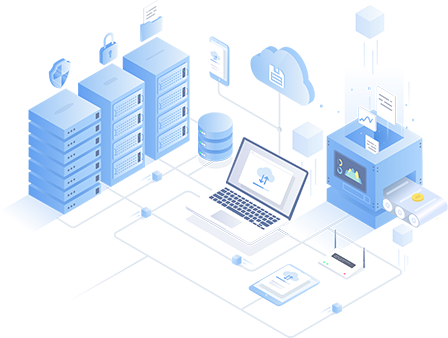
轻蜂加速器全球62个国家中的数百台高速服务器 安全稳定的VPN协议

轻蜂加速器随时随地获得快速稳定的连接
轻蜂加速器遍布各地高速服务器,轻蜂加速器将随时随地为您带来最佳体验。

轻蜂加速器数据加密和隐私保护
轻蜂加速器专注于保护用户的隐私。通过各种加密算法,没有人可以窥探您的Internet活动

轻蜂加速器技术支持
您可以直接与轻蜂加速器的开发人员联系以寻求帮助。我们的技术人员将帮助您解决所有问题。

轻蜂加速器保护您的所有设备
轻蜂加速器即刻享受Windows,IOS, Android精美无障碍的客户端,轻蜂加速器给您完美体验连接只需要一个开关即可,访问网络,只是片刻之间
轻蜂加速app下载适用于:







轻蜂加速器
各地精选高速线路覆盖,可以访问你喜欢的网站,隐私加密,安全稳定
 Windows下载
Windows下载 IOS下载
IOS下载 Android下载
Android下载 MAC下载
MAC下载